In line with the Twelve Principles of Circular Hydrometallurgy (Binnemans & Jones, Sustain. Metall. 9, 1–25 (2023)), SOLVOMET develops solvent extraction process to regenerate the acids that are used for the leaching of (critical) metals from a multitude of input streams. In this new work – in the framework of the Horizon Europe ENICON project – such solvent extraction processes were complemented with the development of a unique molecular thermodynamic model. This work is part of SOLVOMET’s ambition to develop predictive thermodynamic modelling frameworks, which have the advantage that they (1) can (partly) replace inefficient trial-and-error-based research, (2) provide an indispensable step towards rigorous process control, and (3) deliver chemical insights into hydrometallurgical processes. The work was published in Separation and Purification Technology.
Why recovery of acids from aqueous streams?
Recovery of acids from aqueous streams is essential in a broad range of chemical and metallurgical processes. Examples include acid removal from waste solutions to manage environmental concerns, acid recycling to lower the consumption of chemicals, and acidity control during or in between unit operations. Solvent extraction is an interesting technique for acid recovery. It avoids the large chemical consumption and waste generation typical for the neutralization of acids by addition of alkali.
Paper Abstract
Removal or recovery of acids by solvent extraction is highly relevant for recycling, process control, and wastewater decontamination, especially in the hydrometallurgical industry. The construction and optimization of such processes would benefit from a molecular thermodynamic model that can predict liquid–liquid equilibria.
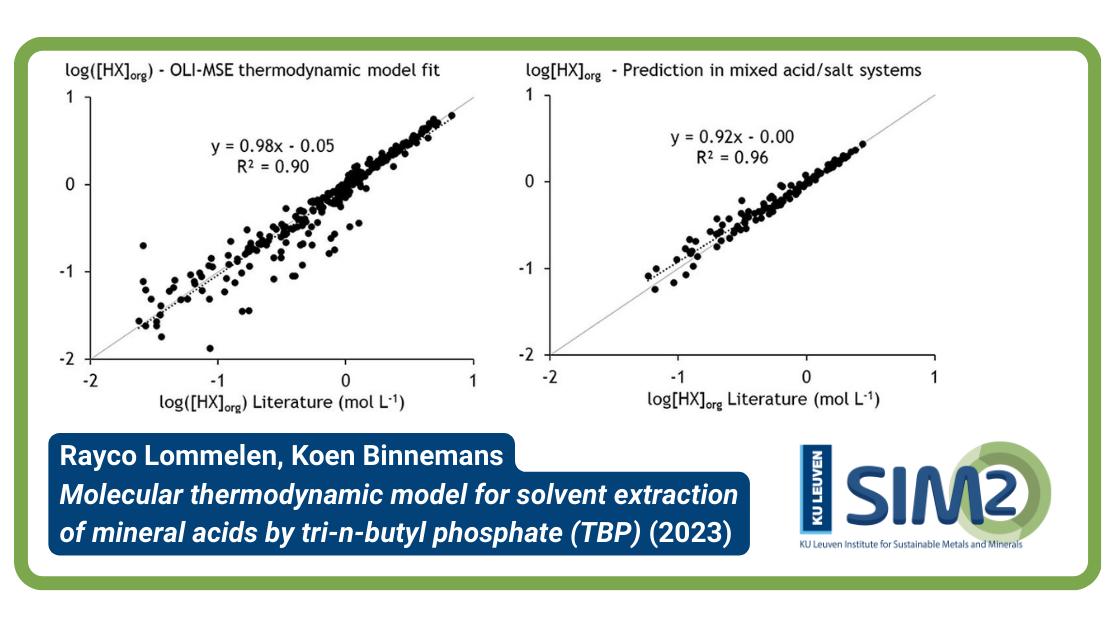
Past attempts resulted in models that were not very predictive. Therefore, a new approach was followed and demonstrated for the extraction of the mineral acids (inorganic acids) HNO3, HCl, H2SO4, H3PO4, and H3AsO4 by tri–n–butyl phosphate (TBP). The semi-empirical OLI Mixed-Solvent Electrolyte (MSE) framework was used to construct the thermodynamic model for calculating the liquid–liquid equilibria.
Contrary to previous attempts, this framework allows the description of both the aqueous and organic phases in one model, and it can accurately deal with the non-ideal behavior of concentrated electrolyte solutions. The best agreement between calculated and experimental distribution data was achieved by assuming that the extraction of mineral acids occurs via protonation of TBP and coextraction of the anions.
At very high acid concentrations, also neutral acid molecules are transferred to the organic phase. The high accuracy of the thermodynamic model for all mineral acid systems considered in this study is an indication that this approach for modeling liquid–liquid equilibria is a universal one.
Furthermore, the extraction of mineral acids can be predicted in mixed-acid systems and acid-salt systems that were not used to construct the model.
Full reference of the paper
Rayco Lommelen, Koen Binnemans, Molecular thermodynamic model for solvent extraction of mineral acids by tri-n-butyl phosphate (TBP), Separation and Purification Technology, Volume 313, 2023,123475, ISSN 1383-5866, https://doi.org/10.1016/j.seppur.2023.123475. (https://www.sciencedirect.com/science/article/pii/S1383586623003830)
Acknowledgments
This work was supported by the European Union’s Framework Programme for Research and Innovation Horizon Europe [Grant number 101058124 (ENICON)]. The authors want to thank Jan Luyten and Joris Roosen (Umicore, Belgium) for the insightful discussions during the construction of the thermodynamic model.